Case study
Selecting the best HCP-ELISA kit out of five
Comparing the coverage and ability to detect HCPs of concern of commercial kits
Selecting the best HCP-ELISA for use during early drug development is crucial: Suppose the chosen ELISA has insufficient coverage of host cell protein (HCP) impurities in the final product. In that case, it may result in project delays due to new assay development and purification improvements required by regulatory authorities.
Our client is an international biotech company developing antibody therapeutics for oncology treatment. They came to us, asking for a comparison of the performance of five commercial CHO kits from four suppliers.
The method applied for this study was ELISA-MS™, which combines immunocapture and mass spectrometry for HCP coverage analysis. A benefit of ELISA-MS™ is that it does not require a mock sample but works with early and complex process samples. Moreover, the method uses less than 1 mg of antibodies. This is much more cost-efficient than techniques based on affinity purification columns, which often need 10-15 mg ELISA antibodies.
HCP ELISA coverage analysis on harvest samples
To find the best ELISA kit for our client’s biologic, we analyzed the HCP content of a harvest cell culture fluid (HCCF) sample supplied by the client. We then compared the coverage of the five commercial HCP-ELISA kits. The figure below shows the coverage of each kit. As you can see, kits A, D, and E each had relatively high coverage, above 60 percent.
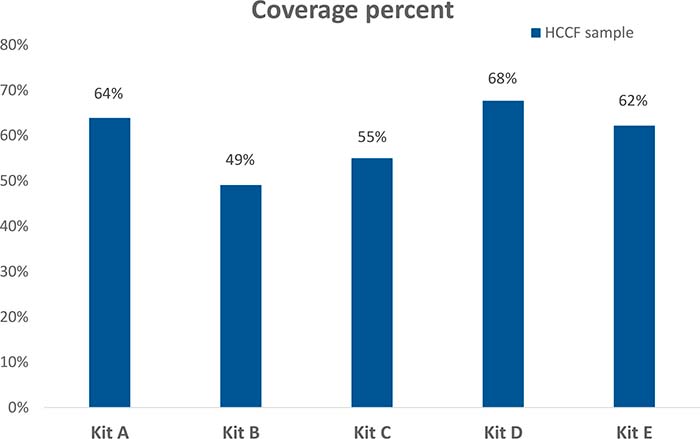
Comparison of Host Cell Proteins in an early process sample covered by five ELISA kits
Detection of Host Cell Proteins of concern
Our client could have based their choice of ELISA on total HCP coverage. However, they also wanted to evaluate the kits’ ability to detect individual HCPs of concern. There were five HCPs on their watch list, and we investigated how well the five ELISAs covered them.
In the table below, green indicates a covered HCP. Our results showed that kit A had the best coverage of the five specific HCPs of concern. This information, in conjunction with the coverage percentages, allowed our client to choose the best commercial ELISA kit for their product.
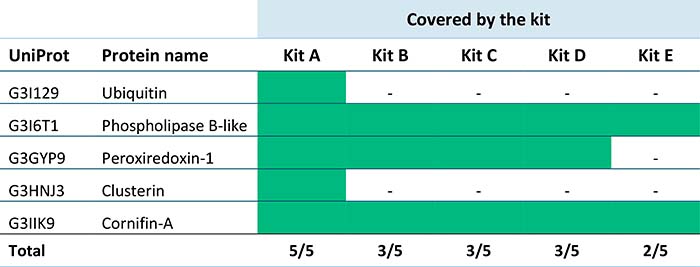
Evaluation of how well five kits detected Host Cell Proteins of concern in a harvest sample
Talk to us
Whatever protein-related challenge or question you may have, we would love to help. Our experts can help you decide on the best analytical approach for your project by email or online meeting - providing advice without obligation.